48 |
Oilfield Technology
September 2016
Enhanced oil recovery technologies are becoming more
commonplace, even in low oil price environments, since they
can significantly reduce water cuts and achieve higher recovery
factors compared to conventional flooding. Steam and alkaline
flooding have proven to be highly effective tertiary oil recovery
methods, enabling the highest possible recovery rates of an oil
reservoir. However, silicate (and carbonate) scale deposition,
associated with long-term steam and alkaline flooding, has been
a major challenge in the adoption of this technology in the oil
and gas industry, typically manifested by steam generator failure,
formation damage, and injection/production well blockage.
Commonlyusedsilicascalepreventionmethods
Various methods can be used to prevent mineral scale formation,
including:
Ì
Operate system at low super-saturation.
Ì
Remove hardness via ion exchange.
Ì
Control pH by acid feed.
Ì
Use scale inhibitors.
Adding a scale inhibitor is generally considered the most
cost-effective scale prevention technique. Inhibitors efficiently
control inorganic scale species, such as calcium carbonate,
calcium sulfate, strontium sulfate, and barium sulfate. However,
silica scale (SiO
2
) is a covalent amorphous solid that cannot be
effectively inhibited by commonly used anionic scale inhibitor
chemistries; neither can it be easily mimicked in laboratory
tests due to its polymorphous nature. Thus, controlling silica
precipitation and deposition is a challenge.
In order to mitigate these challenges, the silica formation
mechanism needs first to be understood. The formation of silica
scale can be described as a three-step process. The first step
consists of the dissolution of silicon dioxide (like quartz), which
results in the formation of silicic acid. Silicic acid itself already
describes a family of compounds containing the element silicon
attached to oxide and hydroxyl groups (Figure 1 illustrates
the most common form of silicic acid) indicating the chemical
complexity of siliceous compounds.
Chemical activation is required for the formation of silicates
and is provided by a hydroxyl anion, which creates a negative
charge on one of the silicate oxygen atoms (illustrated in Figure 2).
And last, the polymerisation reaction of silicates with silicic
acids. Due to the huge variety of available hydroxyl functions,
polymerisation can occur in many directions, which ultimately
yields a large variety of polymorphous colloidal silica products.
This inter- and intramolecular polymerisation is sensitive to the
actual conditions in a production system or a reservoir.
Amorphous silica polymerisation follows a pattern of
soluble silica unit breakdown and silicate
anion growth into larger molecular
networks.
Looking at the silica formation
mechanism, it is clear that the driver for
silica polymerisation is dominated by
silica dissolution in water and formation of
hydroxyl anions. Thus, formation of silica
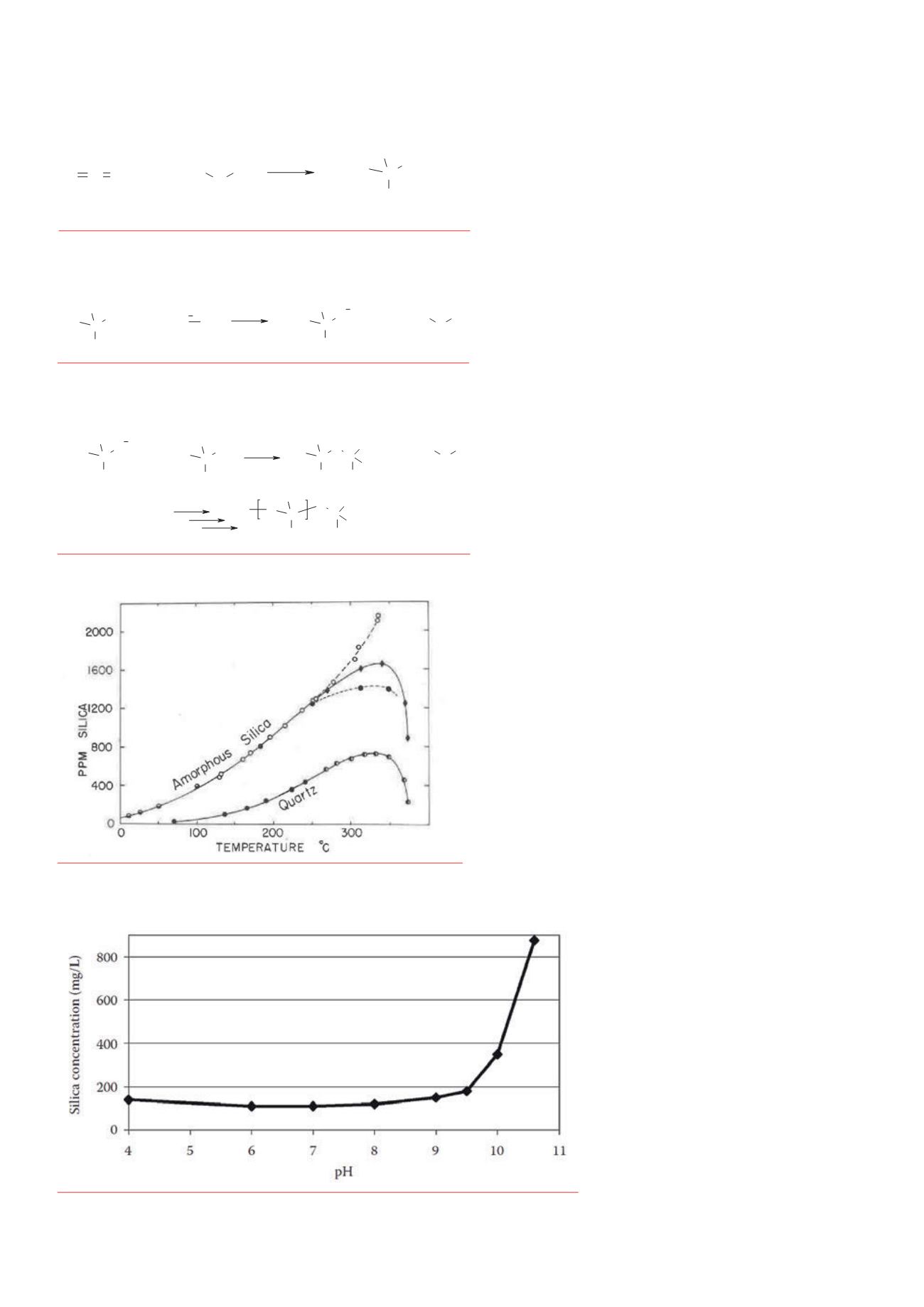
scale is influenced by two major physical
measures: temperature and pH. The
solubility product of amorphous silica and
quartz is strongly related to temperature.
The higher the temperature, the more silica
is dissolved in water, as shown in Figure 4.
Also, as illustrated in Figure 5, the
dissolution of silica increases exponentially at
pH levels of 9 and above.
Figure 2.
Reactionwithactive hydroxyl anions to formsilicates.
Page 2 of 7
Step 1: Silica dissolution in water.
O
H H
Si
O O
Si
OH OH
OH
OH
+
Chemical activation is required for the formation of silicates and is provided by a hydroxyl anion, which
creates a negative charge on one of the silicate oxygen atoms (illustrated in step 2).
Step 2: Reaction with active hydroxyl anions to form silicates.
Si
OH OH
OH
OH
O H
Si
O OH
OH
OH
O
H H
+
+
And last, the polymerisation reaction of silicates with silicic acids. Due to the huge variety of available
hydroxyl functions, polymerisation can occur in many directions, which ultimately yields a large variety
of polymorphous colloidal silica products. This inter- and intramolecular polymerisation is sensitive to
the actual conditions in a production system or a reservoir.
Step 3: Silica polymerisation to form colloidal silica
Si
O OH
OH
OH
Si
OH OH
OH
OH
Si
O OH
OH
OH
Si
OH
OH
OH
O
H H
+
+
Si
O O
OH
OH
Si
OH
OH
OH
H
n
Amorphous silica polymerisation follows a pattern of soluble silica unit breakdown and silicate anion
growth into larger molecular networks.
Looking at the silica formation mechanism, it is clear that the driver for silica polymerisation is
dominated by silica dissolution in water and formation of hydroxyl anions. Thus, formation of silica
scale is influenced by two major physical measures: temperature and pH. The solubility product of
amorphous silica and quartz is strongly related to temperature. The higher the temperature, the more
silica is dissolved in water, as shown in Figure 1.
Page 2 of 7
Step 1: Silica dissolution in water.
O
H H
Si
O O
Si
OH OH
OH
OH
+
Chemical activation is required for the formation of silicates and is provided by a hydroxyl anion, which
creates a negative charge on one of the silicate oxygen atoms (illustrated in step 2).
Step 2: R action with active hydroxyl anions to form silicate .
Si
OH OH
OH
OH
O H
Si
O OH
OH
OH
O
H H
+
+
And last, the polymerisation reaction of silicates with silicic acids. Due to the huge variety of available
hydroxyl functions, polymerisation can occur in many directions, which ultimately yields a large variety
of polymorphous colloidal silica products. This inter- and intramolecular polymerisation is sensitive to
the actual conditions in a production system or a reservoir.
Step 3: Silica polymerisation to form colloidal silica
Si
O OH
OH
OH
Si
OH OH
OH
OH
Si
O OH
OH
OH
Si
OH
OH
OH
O
H H
+
+
Si
O O
OH
OH
Si
OH
OH
OH
H
n
Amorphous silica polymerisation follows a pattern of soluble silica unit breakdown and silicate anion
growth into larger molecular networks.
Looking at the silica formation mechanism, it is clear that the driver for silica polymerisation is
dominated by silica dissolution in water and formation of hydroxyl anions. Thus, formation of silica
scale is influenced by two major physical measures: temperature and pH. The solubility product of
amorphous silica and quartz is strongly related to temperature. The higher the temperature, the more
silica is dissolved in water, as shown in Figure 1.
Figure 3.
Silicapolymerisation to formcolloidal silica.
Figure 1: Dissolution of silica compounds as a function of temperature (see Fournier and Rowe, Amer.
Mineralogist, 62, 1052 -1056, 1977
Also, pH has a significant impact on the formation of silica scale. As illust ate i Figur 2, the
dissolution of silica increases exponentially at pH levels of 9 and above.
Figure 2: Dissolution of silica compounds as a function of pH (Tang and Su-Fen, 1980)
Predicting silica scale
As mentioned above, the formation of silica scale can be described as a function of temperature and
Figure 4.
Diss lution of silica compounds as a function of t mperature
(Fournier andRowe, Amer. Mineralogist, 62, 1052, 1977).
Figure 5.
Dissolution f silica comp unds as a function of pH (Tangand Su-Fen, 1980).
1: Silica dissolution in water.
O
H H
Si
O O
Si
OH OH
OH
OH
+
ical activation is required for the formation of silicates and is provided by a hydroxyl anion, which
es a negative charge on one of the silicate oxygen atoms (illustrated in step 2).
2: Reaction with active hydroxyl ani ns to form silicates.
Si
OH
OH
OH
O H
i
O OH
OH
O
H
+
+
last, the polymerisation reaction of silicates with silicic acids. Due to the huge variety of available
xyl functions, polymerisation can occur in many directions, which ultimately yields a large variety
lymorphous colloidal silica products. This inter- nd intramolecular polymerisation is sensitive to
ctual conditions in a production system or a reservoir.
3: Silica polymerisation to form colloidal silica
Si
O
OH
OH
Si
OH OH
OH
OH
Si
O OH
OH
OH
Si
OH
OH
OH
O
H H
+
+
Si
O O
OH
OH
Si
OH
OH
OH
H
n
phous silica polymerisation foll ws a pattern of soluble silica u it breakdown and silicate anion
th into larger olecular networks.
ing at the silica formation mechanism, it is clear that the driv r for silica polymerisation is
nated by ilica i solut on in water and fo mation of hydroxyl anions. Thus, formation of silica
is influenced by two major physical measures: temperature and pH. The solubility product of
phous silica and quartz is strongly related to temperature. The higher the temperature, the more
is dissolved in water, as shown in Figure 1.
Figure 1.
Silicadissolution inwater.


